ClinShield

Most drugs die before human trials. Not yours.
ClinShield – Automated regulatory analysis for drug development
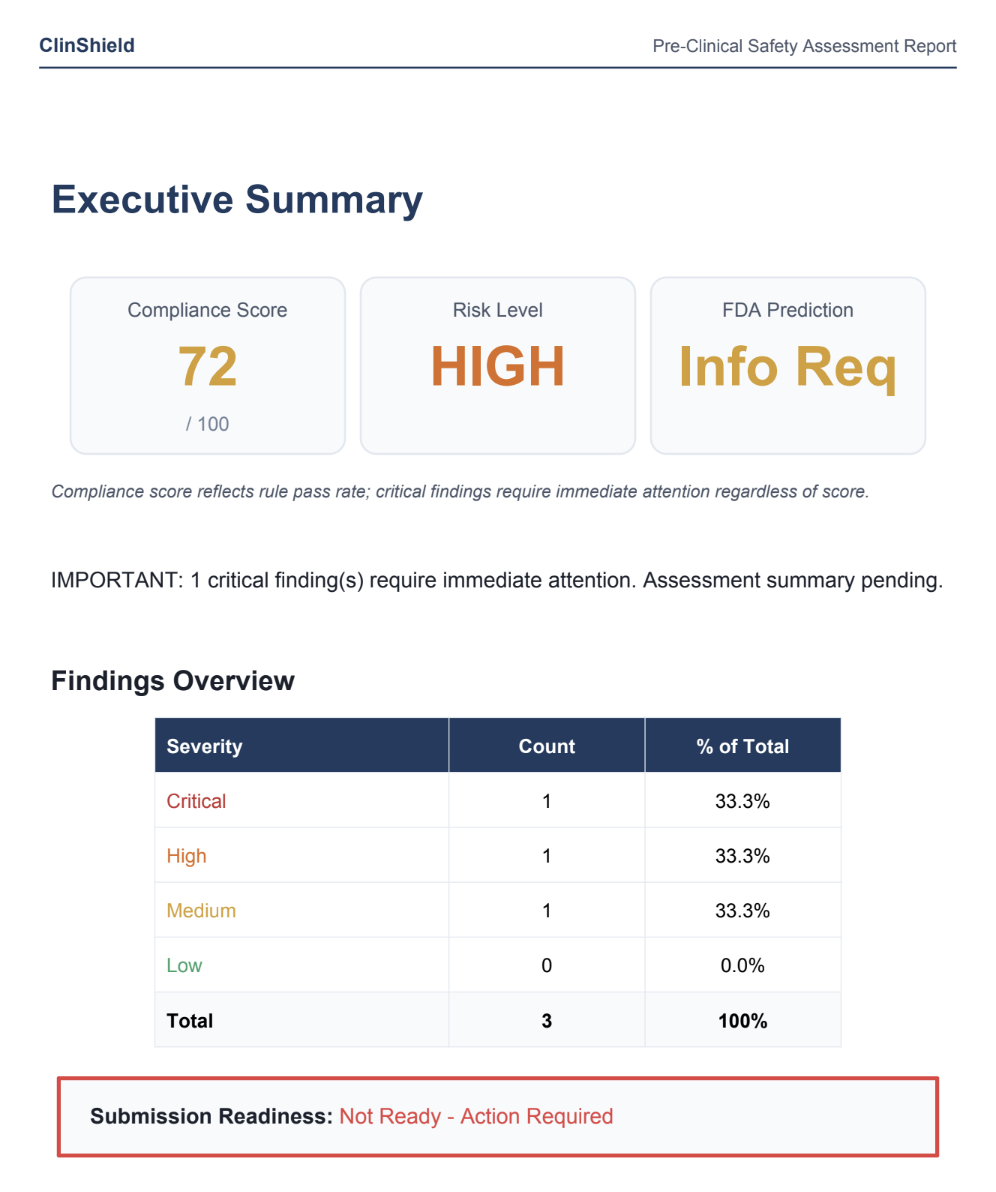
Summary: ClinShield automates regulatory analysis for pre-clinical drug studies by checking over 200 FDA and ICH rules, calculating MRSD, and generating submission-ready reports. It reduces the time and cost of preparing for human trials by replacing manual consultant work.
What it does
Users upload pre-clinical data to receive risk-scored findings with citations, MRSD calculations, and a report aligned with regulatory guidelines. This process, previously taking months and high costs, is completed in minutes.
Who it's for
ClinShield targets pharma, biotech, and CRO professionals involved in IND programs and drug regulatory submissions.
Why it matters
It prevents costly 12-18 month delays caused by Clinical Holds through instant, comprehensive regulatory compliance checks.
